Starting with thorium-234, this nucleus ejects a beta particle, so we go ahead and putĪ beta particle in here, so zero and negative one, If a beta particle isĮjected from the nucleus of a thorium-234, so we're We could put a beta here,Īnd it's an electron, so a negative one charge,Īnd then a zero here.

So here's our electron and an electron ejected from the nucleus We saw in the previous video that you represent an electron, since it has a negative one charge, you put a negative one down here, it's not a proton, nor is it a neutron, so we put a zero here. So this is just a visual representation of what's going on here, To eject an alpha particle, so an alpha particle isĮjected from this nucleus, so we're losing this alpha particle, and what's left behind Happening visually, we're starting off with a uranium nucleus which is unstable, it's going The identity of the other product, just look it up here at our table, find atomic number of 90, and you'll see that's thorium here. We already have two positive charges from our alpha particle, and so we need 90 more. On the left, I know I have 92 protons, so 92 positive charges on the left. In terms of charge, I knowĬharge is also conserved. Total of 238 on the right, and so therefore nucleonsĪre conserved here. Well, I have four from my alpha particle, so I need 234 more.

Trying to figure out the other product from our nuclear equation, I know nucleons are conserved, so if I have 238 nucleons on the left, I need 238 nucleons on the right. So for representing anĪlpha particle in our nuclear equation, since an alpha particle has the same compositionĪs a helium nucleus, we put an He in here, and it has two positive charges, so we put a two down here, and then a total of four nucleons, so we put a four here.
Since there are two protons, the charge of an alpha There are two protons in the helium nucleus and two neutrons. An alpha particle has the same composition as a helium nucleus.
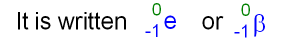
In alpha decay, an alpha particle is ejected from an unstable nucleus, so here's our unstable Many centimetres of lead or many meters of concrete are required to absorb high levels of gamma rays.- Let's look at three types of radioactive decay, and we'll start with alpha decay. It cannot be said that a particular thickness of a material can absorb all gamma radiation. Gamma rays are highly energetic waves and are poor at ionising other atoms or molecules. Gamma rays are the most penetrating of the radiations. Beta particles can be stopped by a few millimetres of aluminium. In a sheet of paper the molecules are much close together so the penetration of alpha particles is much less than in air.īeta particles travel faster than alpha particles and carry less charge (one electron compared to the 2 protons of an alpha particle) and so interact less readily with the atoms and molecules of the material through which they pass. With each collision they lose some of their energy in ionising the air molecule until eventually they give up all of their energy and are absorbed. As alpha particles travel through air they collide with nitrogen and oxygen molecules. Alpha particles can be absorbed by a thin sheet of paper or by a few centimetres of air.
